AG Dr. Fuchs
Members
Michael Fuchs
PhD student
MD Student
The project “Super-resolution force spectroscopy to delineate early pathogenic effects of autoantibody and cytokines in desmosome dysfunction” is funded by the DFG Collaborative Research Center TRR 425. Our goal is to dissect the early spatiotemporal sequence of events triggered by pathogenic stimuli that ultimately lead to loss of desmosomal adhesion. We focus on two disease models: Pemphigus vulgaris (PV) in the skin and inflammatory bowel diseases (IBD) in intestinal epithelial cells. In addition to biochemical approaches, we apply state-of-the art imaging and biophysical techniques, including stimulated emission depletion (STED) microscopy, atomic force microscopy (AFM), and their combined application (STED/AFM), to analyze desmosomal adhesion upon pathogenic stimuli at a single molecule level (Figure 1). By utilizing our correlative STED/AFM setup and a murine keratinocyte cell line stably expressing desmoplakin-eYFP, we established a platform that enables precise force spectroscopy measurements within individual desmosomes.

Figure 1; modified form Fuchs et al., CMLS, 2023.
PV is a severe autoimmune skin disease in which autoantibodies target adhesion-mediating desmosomal proteins, primarily desmoglein (Dsg) 1 and Dsg3. Affected patients develop severe skin blistering, which, if untreated, can be life-threatening. Two principal pathomechanisms have been proposed: Steric hindrance of desmoglein interactions and alteration of intracellular signaling pathways. Both mechanisms comprise desmosomal adhesion. We previously showed that under cell culture conditions, steric hindrance precedes activation of p38MAPK signaling, with both mechanisms contributing distinctly to loss of adhesion (Figure 2). Similar mechanisms have been described in inflammatory bowel disease (IBD), where pro-inflammatory cytokines are thought to impair Dsg2-mediated adhesion through activation of p38MAPK signaling.
Given the similarity of these pathomechanisms, we use both disease models to investigate the precise spatial and temporal sequence of molecular events, as well as the interplay between steric hindrance and altered signaling pathways following pathological stimulation. By integrating super-resolution imaging with force spectroscopy, we determine how individual molecular alterations, depending on their localization, contribute to the loss of desmosomal adhesion. Furthermore, we aim to evaluate the mechanisms of potential therapeutic components, such as Apremilast and the Tandem Peptide, that may stabilize desmosomal adhesion.
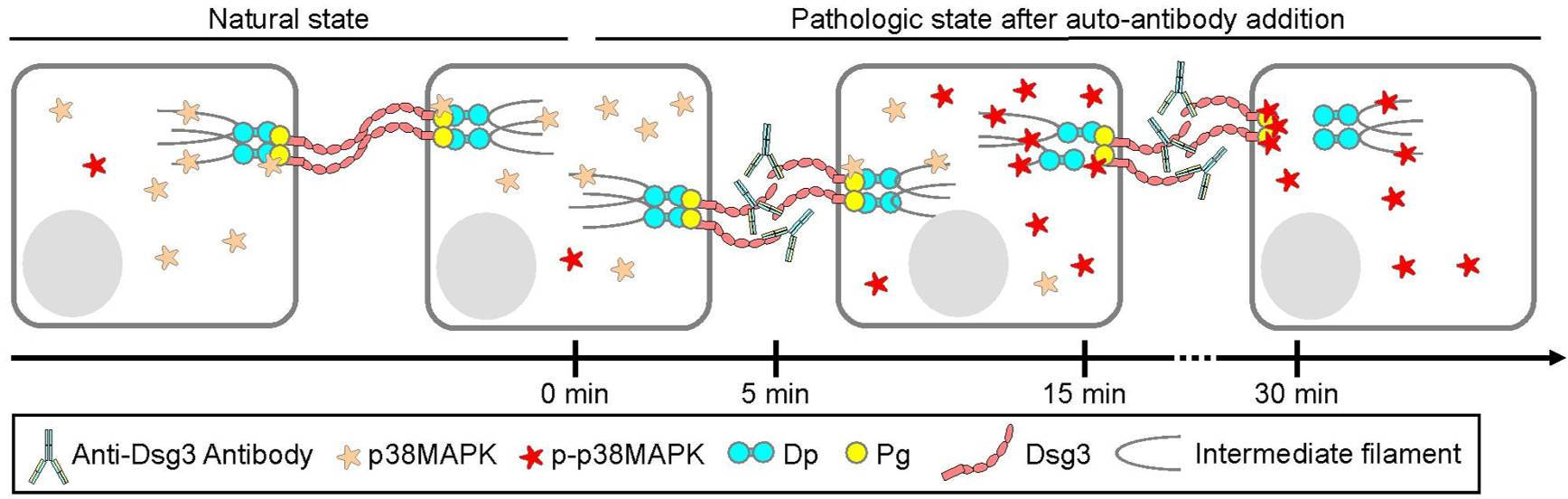
Figure 2; modified from Fuchs et al., BJD, 2025.
Publications
Fuchs M, Möchel M, Radeva MY, Schmitt T, Yazdi AS, Hashimoto T, Waschke J. In desmosomes direct inhibition precedes p38MAPK-mediated uncoupling to reduce Dsg3 adhesion by pemphigus autoantibodies. Br J Dermatol. 2025 Apr 23. doi: 10.1093/bjd/ljaf142.
Egu DT, Schmitt T, Ernst N, Ludwig RJ, Fuchs M, Hiermaier M, Moztarzadeh S, Morón CS, Schmidt E, Beyersdorfer V, Spindler V, Steinert LS, Vielmuth F, Sigmund AM, Waschke J. EGFR Inhibition by Erlotinib Rescues Desmosome Ultrastructure and Keratin Anchorage and Protects against Pemphigus Vulgaris IgG-Induced Acantholysis in Human Epidermis. J Invest Dermatol. 2024 Nov;144(11):2440-2452. doi: 10.1016/j.jid.2024.03.040.
Steinert L, Fuchs M, Sigmund AM, Didona D, Hudemann C, Möbs C, Hertl M, Hashimoto T, Waschke J, Vielmuth F (2024). Desmosomal hyper-adhesion affects direct inhibition of desmoglein interactions in pemphigus. Journal of Investigative Dermatology, 144(12). doi: 10.1016/j.jid.2024.03.042
Fuchs, M., Radeva, M. Y., Spindler, V., Vielmuth, F., Kugelmann, D., and Waschke, J. (2023). Cytoskeletal anchorage of different Dsg3 pools revealed by combination of hybrid STED/AFM. Cellular and Molecular Life Sciences 80(1), 25. doi: 10.1007/s00018-022-04681-9
Sigmund AM, Winkler M, Engelmayer S, Kugelmann D, Egu DT, Steinert LS, Fuchs M, Hiermaier M, Radeva MY, Bayerbach FC, Butz E, Kotschi S, Hudemann C, Hertl M, Yeruva S, Schmidt E, Yazdi AS, Ghoreschi K, Vielmuth F, Waschke J. Apremilast prevents blistering in human epidermis and stabilizes keratinocyte adhesion in pemphigus. Nat Commun. 2023 Jan 9;14(1):116. doi: 10.1038/s41467-022-35741-0.
Fuchs M, Kugelmann D, Schlegel N, Vielmuth F, Waschke J. Desmoglein 2 can undergo Ca2+-dependent interactions with both desmosomal and classical cadherins including E-cadherin and N-cadherin. Biophys J. 2022 Apr 5;121(7):1322-1335. doi: 10.1016/j.bpj.2022.02.023.
Fuchs M, Sigmund AM, Waschke J, Vielmuth F. Desmosomal Hyperadhesion Is Accompanied with Enhanced Binding Strength of Desmoglein 3 Molecules. Biophys J. 2020 Oct 20;119(8):1489-1500. doi: 10.1016/j.bpj.2020.09.008.
Kugelmann D, Rötzer V, Walter E, Egu DT, Fuchs MT, Vielmuth F, Vargas-Robles H, Schnoor M, Hertl M, Eming R, Rottner K, Schmidt A, Spindler V, Waschke J. Role of Src and Cortactin in Pemphigus Skin Blistering. Front Immunol. 2019 Apr 4;10:626. doi: 10.3389/fimmu.2019.00626.
Fuchs M, Foresti M, Radeva MY, Kugelmann D, Keil R, Hatzfeld M, Spindler V, Waschke J, Vielmuth F. Plakophilin 1 but not plakophilin 3 regulates desmoglein clustering. Cell Mol Life Sci. 2019 Sep;76(17):3465-3476. doi: 10.1007/s00018-019-03083-8.
Vielmuth F, Walter E, Fuchs M, Radeva MY, Buechau F, Magin TM, Spindler V, Waschke J (2018). Keratins regulate p38MAPK-dependent desmoglein binding properties in pemphigus. Frontiers in Immunology, 9, 528. Doi: 10.3389/fimmu.2018.00528.

