AG Dr. Schmitt
Members
Group leader: Dr. rer. nat. Thomas Schmitt
Technician: Martina Hitzenbichler
PhD student: T.B.D.
Desmososomes and Pemphigus
Desmosomes are specialized cell–cell junctions that provide strong mechanical adhesion between cells. They are composed of the cadherins desmogleins (Dsg) and desmocollins (Dsc), which are anchored intracellularly by plaque proteins such as plakoglobin (Pg), plakophilins (Pkp), and desmoplakin (Dp). Through their connection to the keratin cytoskeleton, desmosomes ensure tissue cohesion and resistance to mechanical stress for example in epithelia.
Pemphigus is a severe autoimmune blistering disease in which autoantibodies target desmogleins, most commonly Dsg1 and Dsg3 in pemphigus vulgaris (PV). Autoantibody binding not only interferes with adhesive interactions but also activates intracellular signaling pathways that destabilize desmosomes. The resulting loss of cell adhesion leads to fluid-filled blisters of the skin and mucous membranes (Figure 1). If untreated, the disease can be life-threatening due to barrier failure resulting in fluid loss, and infections.
Current therapies rely mainly on immunosuppression. Although often effective, these treatments can cause significant side effects and may act slowly. Our research therefore aims to better understand the mechanisms regulating desmosome adhesion in order to identify strategies that directly stabilize cell–cell adhesion and provide faster, more targeted therapeutic options.

Figure 1: Hematoxylin–eosin staining of a PV erosion in patient skin. Schmitt, Waschke; Frontiers in Medicine; 2021 DOI: 10.3389/fmed.2021.701809
Desmosome ultrastructure in health and disease
One focus of our work is the analysis of desmosome ultrastructure and how structural alterations contribute to adhesion loss in pemphigus. We have identified several characteristic changes associated with impaired adhesion, including:
• Retraction of keratin filaments from the desmosomal plaque
• Reduced desmosome number
• Internalization of desmosomal components
• Reduced desmosome size and altered plaque architecture
Using advanced imaging techniques such as stimulated emission depletion (STED) super-resolution microscopy, we investigate how these structural changes relate to distinct adhesive states and how they are influenced by specific signaling pathways (Figure 2).
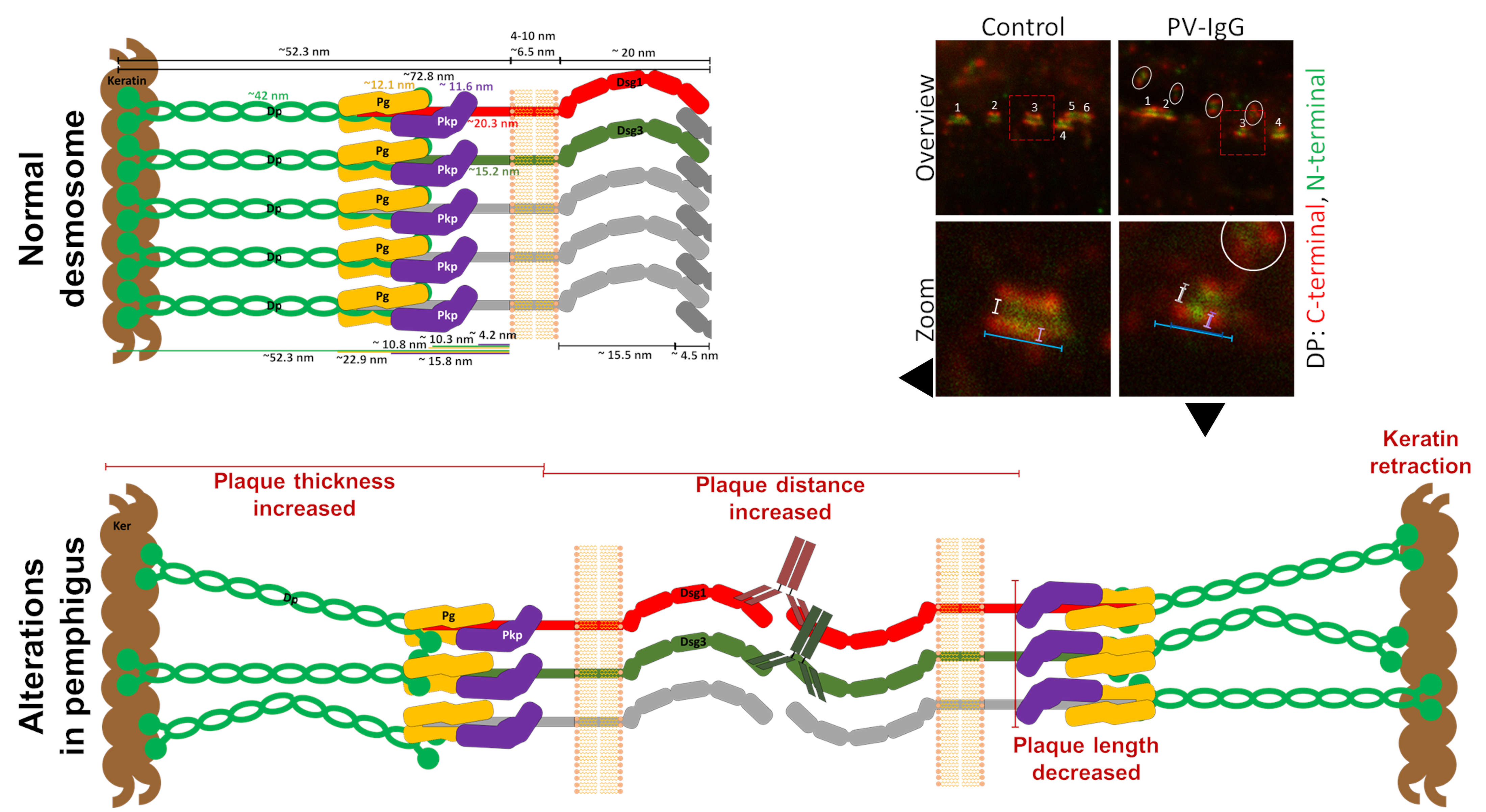
Figure 2: Desmosome ultrastructure and alterations induced by pemphigus vulgaris IgG (PV-IgG).Effects include: a decreased number of desmosomes (white numbers), internalization of desmosomes (white circles), an increased distance between the C-terminal domains of desmoplakin (Dp) in the two desmosomal halves (white/grey spans), decreased desmosome size (blue spans), and increased Dp plaque thickness (magenta spans). Dsg1 or 3: desmoglein 1 or 3; Dsc: desmocollin; Pg: plakoglobin; Pkp: plakophilin.
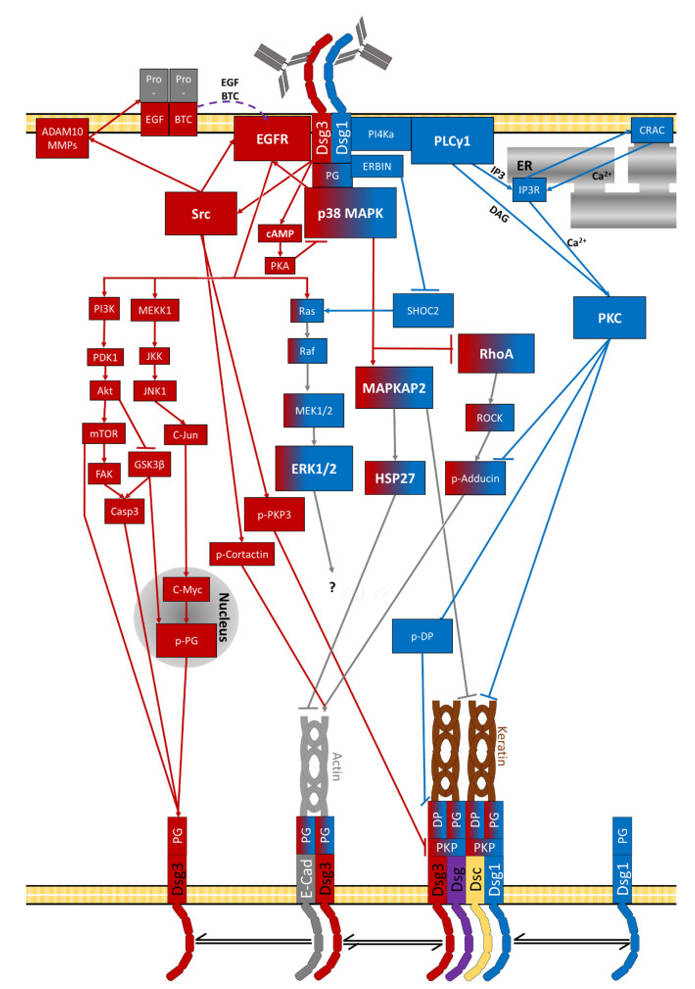
Deciphering the Pemphigus Signaling Network
A second major focus of our research is the signaling network activated downstream of autoantibody binding. Through biochemical and functional assays, we analyze which signaling pathways are activated or inhibited and how they interact to regulate desmosome stability.
Our work has demonstrated that anti-Dsg1 and anti-Dsg3 antibodies trigger distinct signaling responses. Furthermore, epitope-specific anti-Dsg3 antibodies differentially affect disease phenotype and may influence disease severity and treatment response. Identifying key signaling nodes and specific downstream targets may open new avenues for targeted therapy (Figure 3).
Funding
This work is funded by the Deutsche Forschungsgemeinschaft (DFG) within the Collaborative Research Center SFB/TRR425 DEFINE (Project A02).
Most relevant publications
T. Schmitt, D. T. Egu, E. Walter, A. M. Sigmund, R. Eichkorn, A. Yazdi, E. Schmidt, M. Sárdy, R. Eming, M. Goebeler, J. Waschke: Ca2+ signalling is critical for autoantibody-induced blistering of human epidermis in pemphigus, The British journal of dermatology, 2021. DOI: https://doi.org/10.1111/bjd.20091
C. Hudemann, R. Maglie, M. Llamazares-Prada, B. Beckert, D. Didona, R. Tikkanen, T. Schmitt, T. Hashimoto, J. Waschke, M. Hertl, R. Eming: Human Desmocollin 3‒Specific IgG Antibodies Are Pathogenic in a Humanized HLA Class II Transgenic Mouse Model of Pemphigus, Journal of Investigative Dermatology, 2021. DOI: https://doi.org/10.1016/j.jid.2021.06.017
T. Schmitt, J. Pircher, L. Steinert, K. Meier, K. Ghoreschi, F. Vielmuth, D. Kugelmann, J. Waschke: Dsg1 and Dsg3 Composition of Desmosomes Across Human Epidermis and Alterations in Pemphigus Vulgaris Patient Skin, Frontiers in immunology, 25 May 2022; Vol. 13, 884241. DOI: 10.3389/fimmu.2022.884241
D. T. Egu, T. Schmitt, A. M. Sigmund, J. Waschke: Electron microscopy reveals that phospholipase C and Ca2+ signaling regulate keratin filament uncoupling from desmosomes in Pemphigus, Annals of anatomy = Anatomischer Anzeiger official organ of the Anatomische Gesellschaft, 2022; Vol. 241, 151904. DOI: https://doi.org/10.1016/j.aanat.2022.151904
T. Schmitt, C. Hudemann, S. Moztarzadeh, M. Hertl,R. Tikkanen, J. Waschke. Dsg3 epitope-specific signalling in pemphigus, Frontiers in Immunology, Apr 18, 2023. 14:1163066. doi:10.3389/fimmu.2023.1163066
D. T. Egu, T. Schmitt, N. Ernst, R. J. Ludwig, M. Fuchs, M. Hiermaier, S. Moztarzadeh, C. S. Morỏn, E. Schmidt, V. Beyersdorfer, V. Spindler, L. S. Steinert, F. Vielmuth, A. M. Sigmund, J. Waschke, EGFR Inhibition by Erlotinib Rescues Desmosome Ultrastructure and Keratin Anchorage and Protects Against Pemphigus Vulgaris IgG-Induced Acantholysis in Human Epidermis, J Invest Dermatol 2024. DOI: 10.1016/j.jid.2024.03.040
T. Schmitt, J. Huber, J. Pircher, E. Schmidt, J. Waschke. The impact of signaling pathways on the desmosome ultrastructure, Frontiers in Immunology, (Dec. 16., just accapted).
M. Fuchs, M. Möchel, M. Y. Radeva, T. Schmitt, T. Hashimoto, J. Waschke. In desmosomes direct inhibition precedes p38MAPK-mediated uncoupling to reduce Dsg3 adhesion by pemphigus autoantibodies, British Journal of Dermatology, (23.04.2025).
J. Haneberg, T. Schmitt, D. Egu, C. Sebastià, E. Schmidt, C. Hudemann, M. Hertl, T. Hashimoto, J. Waschke. The role of PKC subtype-specific signaling in the regulation of keratinocyte adhesion in pemphigus, British Journal of Dermatology, (15.11.2025).
F. Vielmuth, A. Sigmund, D. Egu, M. Hiermaier, T. Schmitt, L. Steinert, K. Meier, K. Ghoreschi, A. Horn, M. Radeva, D. Kugelmann, J. Waschke. Plakoglobin phosphorylation at serine 665 is required for protective effects of apremilast in pemphigus, JCI Insight (Just accepted).

